Have you ever wondered about the fate of the universe? We have notable evidence to suggest that its birth was due to the Big Bang, but have you ever pondered upon the death of the universe? It’s a terrifying thought to some, but not impossible. In fact, as everything has an end, the universe might have an end as well.
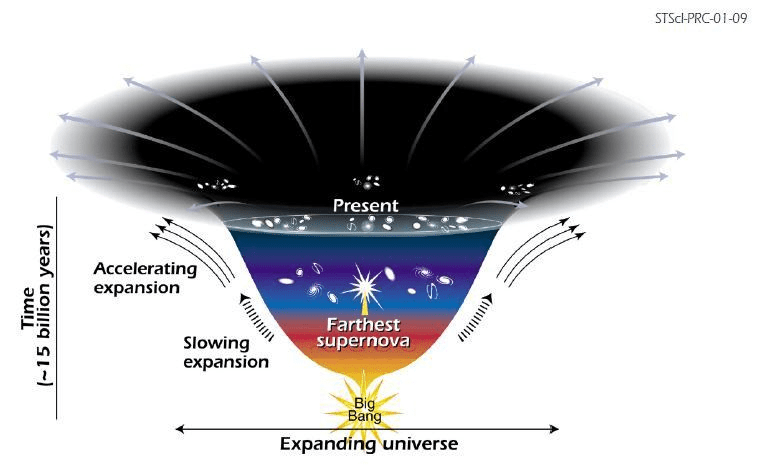
Let’s start this discourse by one of the most famous theories for the ending of the universe : The heat death. In a nutshell, it’s the expanding of the universe and losing heat till it reaches absolute zero (0K) (no molecular motion). To understand the basic idea of it, it’s essential to have a basic idea of the 2nd law of thermodynamics and the definition of entropy.
The 2nd law of thermodynamics was given by Rudolf Claussius and William Thomson, Claussius in particular was credited for defining ‘entropy’. As for Rudolf Claussius, he defined the 2nd law of thermodynamics as ‘a cyclic transformation whose only final result is to transfer heat from a body at a given temperature to a body at a higher temperature is impossible’.
In simpler terms, heat spontaneously flows from hotter to colder parts in a region. This spontaneous action is due to entropy.
Let’s say, here, heat Q flows into a heat reservoir at a constant temperature T. Take the change in entropy as ΔS.
ΔS=Q/T
Now, assume two reservoirs, R1 and R2 at T1 and T2 respectively, so the net change would be
ΔS= Q/T2 -Q/T1
In short, if the entropy is +ve, the action is non-spontaneous and if it’s -ve, it’s spontaneous.

One thing to keep in mind is that entropy is statistical in nature. It only matters when you’re dealing with a large number of molecules interacting with each other. Technically, at molecular level, heat is the random kinetic energy generated due to motion and collisions of molecules. So, now, wouldn’t you ask if it’s impossible for all molecules to collect at one place?
Theoretically, no, but it’s extremely improbable for such an occurrence to occur. And so, we have finally cleared the preface of heat death.
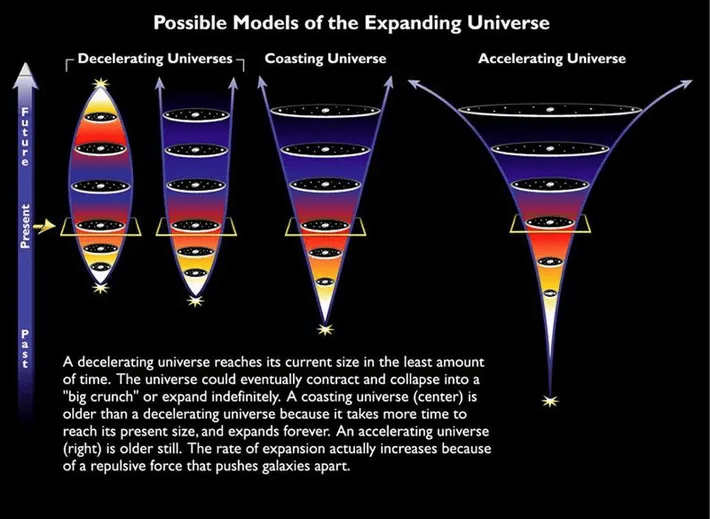
The universe is obviously an isolated system as far as we know. It’s also known that the entropy of the universe is continuously increasing. For heat death to occur, entropy must be at maximum level, for the occurrence of thermal equilibrium. As this is the maximum level of entropy, no further change is possible, which means, no more motion or processes would occur which increase entropy. Chemical processes are needed for life to occur, as according to the 2nd law of thermodynamics, total chemical equilibrium means the end of life.
Although, none of us really need to worry about this, so be relieved as this is not expected to happen till 10¹⁰⁰ years, if it ever happens.

Leave a comment